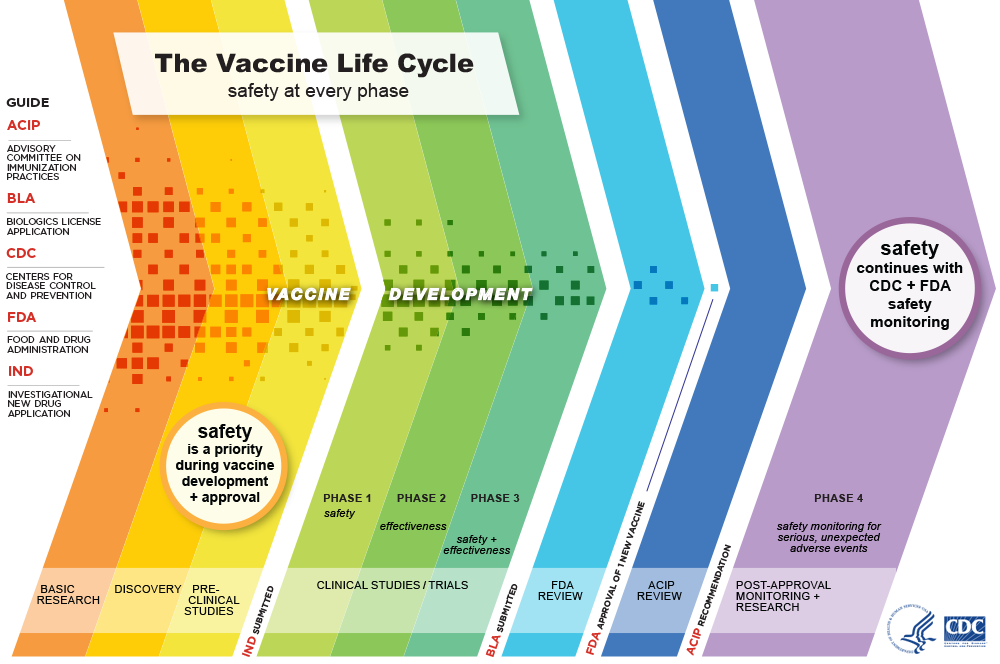
Through decades of research, testing and development, vaccines have proven to be an effective way to prevent many infectious diseases and their effects, including serious illness, hospitalization and death. Vaccines are held to a high safety standard, pass lots of testing and are constantly monitored for safety throughout all stages of use.
Most people do not experience serious side effects from vaccines. Some mild side effects can sometimes occur, such as pain or swelling at the injection site, and these side effects are often signs that a body is building a healthy immune response and protection well.
Getting multiple vaccinations at once has been shown to be safe.
Vaccine Adverse Event Reporting System (VAERS)
It is rare, but an adverse event (possible side effect) can occur after a person receives a vaccine dose. The cause of these adverse events can be difficult to identify and are often from an unrelated cause, not the vaccine. Scientists monitor for immunization response trends and unusual patterns of adverse events. Vaccine Adverse Event Reporting System (VAERS) is used by the Food and Drug Administration (FDA) and Centers for Disease Control and Prevention (CDC) to collect reports of side effects that happen after vaccination. The system relies on individuals to report adverse health events following vaccination – anyone can and should report adverse events to VAERS (hhs.gov).
How to Report an Adverse Event to VAERS
Created in 1990 by the US Department of Health and Human Services, VAERS is a database for the collection of adverse events following vaccines. It is operated and monitored by the CDC and the FDA to detect whether any vaccines are associated with higher than expected rates of adverse events.
Anyone can report to VAERS. This can be vaccine recipients themselves, caregivers of vaccine recipients (parents, guardians) or health care providers. Any clinically significant or unexpected events following a vaccine administration should be reported to VAERS. See VAERS Table of Reportable Events Following Vaccination (PDF).
Adverse events that follow vaccines are rare. Most adverse events that follow vaccines are manageable by health care providers that administer vaccines and will occur within hours of administration. Some extremely rare events can occur within six months of vaccine administration. People with conditions known to make them more vulnerable to these events should talk to their health care provider before receiving a vaccine. Some adverse events and their expected timeline after vaccination are listed here: Vaccine Injury Table Effective for Claims Filed on or After 1-3-2022 (hrsa.gov).
North Dakota Health and Human Services (NDHHS) does not maintain the VAERS database but does regularly review reports made by or on behalf of North Dakotans. To contact the NDHHS Immunization Unit for assistance with or questions about VAERS, please contact vaccine@nd.gov.
How is Vaccine Safety Monitored in the U.S.?
The success of vaccine programs depends on vaccine effectiveness and safety. Because vaccines are given to millions of healthy people each year, they are held to a very high standard and are continuously monitored for safety. The U.S. has one of the most advanced systems in the world for assessing vaccine safety. This includes using state-of-the-art technologies and systems working together. Each of the systems supplies a different type of data for researchers to analyze. Together, they work as a well-oiled machine to help provide a comprehensive picture of vaccine safety in the U.S.
Safety Information by Vaccine
Available Vaccines
Two chickenpox vaccines are approved in the U.S. Both vaccines contain live weakened varicella-zoster virus. One vaccine is a single antigen and the other is a combination vaccine.
Who Should Get Chickenpox Vaccine
Two doses of chickenpox vaccine are recommended by CDC. Children should get the first dose at age 12 to 15 months and the second dose at age 4 to 6 years. The second dose can be given before age 4 to 6 if it is given at least three months after the first dose.
People age 13 years and older who have not gotten chickenpox vaccine and have never had chickenpox should get two doses of chickenpox vaccine. The two doses should be at least 28 days apart.
Common Side Effects
Chickenpox vaccine is safe and the best way to prevent chickenpox. Like any medicine, vaccines can have side effects. The side effects are usually mild and go away on their own.
Common side effects of chickenpox vaccine include:
- Sore arm from shot
- Fever
- Mild rash at spot shot was given
- Temporary pain and stiff joints
Severe allergic reactions to chickenpox vaccine are rare but can be life threatening. In the event of a severe allergic reaction, call 9-1-1 and get the person to the closest hospital.
Who Should Not Get Chickenpox Vaccine
People should not get chickenpox vaccine if they:
- Have had a severe allergic reaction to a past dose of chickenpox vaccine or to any vaccine component
- Have an active infection or fever
- Are or might be pregnant
- Have a weakened immune system due to medication, immune system problems or cancers
- Have untreated active tuberculosis (TB)
Talk to your health care provider about whether or not chickenpox vaccine is right for you.
Available Vaccines
Two types of COVID-19 vaccine are both approved in the U.S. One is mRNA COVID-19 vaccine and one is protein subunit COVID-19 vaccine. Neither type is recommended as the preferred COVID-19 vaccine for protection against COVID-19.
Who Should Get COVID-19 Vaccine
Annual COVID-19 vaccine is recommended by CDC for all people age 6 months and older. Multiple doses of COVID-19 vaccine may be recommended.
Common Side Effects
CODID-19 vaccine is safe and the best way to prevent COVID-19. Like any medicine, vaccines can have side effects. The side effects are usually mild and go away on their own.
Common side effects of COVID-19 vaccine tend to be mild, should go away in a few days and include:
- Pain, swelling and redness at spot shot was given
- Tiredness, headache or muscle pain
- Chills
- Nausea
- Fever
MRNA COVID-19 vaccine can present a rare risk of pericarditis and myocarditis, mostly for males ages 12-39 years. This risk may be decreased by having a longer period of time between the first and second dose of mRNA COVID-19 vaccine. Initial findings show that nearly all patients who have gotten myocarditis from mRNA COVD-19 vaccine have recovered.
Severe allergic reactions to COVID-19 vaccine are rare but can be life threatening. In the event of a severe allergic reaction, call 9-1-1 and get the person to the closest hospital.
Who Should Not Get COVID-19 Vaccine
People should not get COVID-19 vaccine if they:
- Have had a severe allergic reaction to a past dose of COVID-19 vaccine or to any vaccine component
Talk to your health care provider about whether or not COVID-19 vaccine is right for you.
Available Vaccines
- DTap protects children against diphtheria, tetanus and acellular pertussis.
- Tdap protects adolescents and adults against tetanus, diphtheria and acellular pertussis.
- Td protects adolescents and adults from tetanus and diphtheria.
Who Should Get DTap/Tdap/Td Vaccines
Children should receive five doses of DTap vaccine. They should get their first dose at age 2 months, second dose at age 4 months, third dose at age 6 months, fourth dose at age 15-18 months and fifth dose at age 4-6 years.
Children should receive one dose of Tdap at age 11-12 years. People who are pregnant should get one dose of Tdap during each pregnancy, preferably during the early part of their third trimester. Adults who have never gottend Tdap should get one dose of Tdap.
Adults should receive a booster dose of Tdap or Td every 10 years or after 5 years in the case of a severe burn or dirty wound.
Common Side Effects
DTap, Tdap and Td vaccines are safe and the best way to prevent diptheria, tetanus and pertussis (Td vaccine does not prevent pertussis). Like any medicine, vaccines can have side effects. The side effects are usually mild and go away on their own.
Common side effects of DTap vaccine can last 1-3 days and include:
- Sore arm from shot
- Fever
- Fussiness
- Feeling tired
- Loss of appetite
- Vomiting
Common side effects of Tdap include:
- Pain, redness or swelling at spot shot was given
- Mild fever
- Headache
- Feeling tired
- Nausea, vomiting, diarrhea or stomachache
Common side effects of Td vaccine include:
- Pain, redness or swelling at spot shot was given
- Mild fever
- Headache
- Feeling tired
- Nausea, vomiting, diarrhea or stomachache
Severe allergic reactions to DTap, Tdap and Td vaccines are rare but can be life threatening. In the event of a severe allergic reaction, call 9-1-1 and get the person to the closest hospital.
Who Should Not Get DTap/Tdap/Td Vaccines
Children under age 7 years should not get Tdap and Td vaccines.
People should not get DTap/Tdap/Td vaccines if they have had a severe allergic reaction to a past dose of DTap/Tdap/Td vaccines or to any vaccine component, respectively.
Talk to your health care provider about whether or not DTap/Tdap/Td vaccines are right for you.
You can learn more about DTap/Tdap/Td vaccines from the CDC.
Available Vaccines
Five vaccines that protect against Haemophilus influenzae type b (Hib) are approved in the U.S. Three of these are single antigen and two of these are combination vaccines.
Who Should Get Hib Vaccine
Three or four doses of Hib vaccine are recommended by CDC, depending on which vaccine series they get. If getting a 3-dose series, children should get the first dose at age 2 months, second dose at age age 4 months and third dose at age 12-15 months. If getting a 4-dose series, children should get the first dose at 2 months, second dose at age 4 months, third dose at age 6 months and fourth dose at age 12-15 months.
Older children and adults with certain medical conditions should also get Hib vaccine.
Common Side Effects
Hib vaccine is safe and the best way to prevent Hib disease. Like any medicine, vaccines can have side effects. The side effects are usually mild and go away on their own.
Common side effects of single antigen Hib vaccine include:
- Redness, warmth or swelling at spot shot was given
- Fever
- Irritability
Common side effects of combination Hib vaccine include:
- Redness, warmth or swelling at spot shot was given
- Fever
- Irritability
Severe allergic reactions to Hib vaccine are rare but can be life threatening. In the event of a severe allergic reaction, call 9-1-1 and get the person to the closest hospital.
Who Should Not Get Hib Vaccine
People should not get single antigen Hib vaccine if they:
- Have had a severe allergic reaction to a past dose of Hib vaccine or to any vaccine component
- Have had an allergic reaction to latex
People should talk to their health care provider before getting single antigen Hib vaccine if they have a moderate or severe acute illness with or without a fever.
People should not get combination Hib vaccine if they:
- Have had a severe allergic reaction to a past dose of Hib vaccine or any vaccine component
- Developed encephalopathy (such as coma, lowered level of consciousness or prolonged seizures) within seven days of a past dose of DTP or DTaP vaccines
People should talk to their health care provider before getting combination Hib vaccine if they are pregnant, developed GBS (Guillain-Barré syndrome within six weeks of getting a vaccine with tetanus-toxoid or have a history of Arthus-type hypersensitivity reaction after a past dose of vaccine that protects against tetanus or diphtheria.
Talk to your health care provider about whether or not Hib vaccine is right for you.
Available Vaccines
Three vaccines that protect against hepatitis A are approved in the U.S. Two of these are single antigen and one of these is a combination vaccine.
Who Should Get Hep A Vaccine
Two doses of hep A vaccine given at least 6 months apart are recommended by CDC. Children starting at age 1 year, people experiencing homelessness or with unstable housing, people at increased risk for infection, people at increased risk for complications from hep A and any people who want to protect against hep should get hep A vaccine.
Common Side Effects
Hep A vaccine is safe and the best way to prevent hepatitis A. Like any medicine, vaccines can have side effects. The side effects are usually mild and go away on their own.
Common side effects of hep A vaccine include:
- Swelling, tenderness, redness, warmth or a hard lump at spot shot was given
- Low fever
- General ill feeling
- Nausea
- Loss of appetite
- Headache
Severe allergic reactions to Hep A vaccine are rare but can be life threatening. In the event of a severe allergic reaction, call 9-1-1 and get the person to the closest hospital.
Who Should Not Get Hep A Vaccine
People should not get hep A vaccine if they:
- Have had a severe allergic reaction to a past dose of hep A vaccine or to any vaccine component
- Have had an allergic reaction to Neomycin or yeast (contraindication for Twinrix)
People with a minor illness such as a cold can get hep A vaccine but people with a moderate or severe illness should usually wait to get hep A vaccine after they recover.
Talk to your health care provider about whether or not hep A vaccine is right for you.
Available Vaccines
Seven vaccines that protect against hepatitis B are approved in the U.S. Four of these protect against only hepatitis B, one of these protects against both hepatitis A and B and two of these are childhood vaccines that protect against hepatitis B as well as other diseases.
Who Should Get Hep B Vaccine
Two, three or four doses of hep B are recommended by CDC depending on vaccine series and health needs. Infants with 24 hours of birth, children and adolescents less than age 19 years who have not had hep B vaccine, people at greater risk of hep B and any people who want to protect against hep B should get hep B vaccine.
Common Side Effects
Hep B vaccine is safe and the best way to prevent hepatitis B. Like any medicine, vaccines can have side effects. The side effects are usually mild and go away on their own.
Common side effects of hep B vaccine include:
- Pain, soreness, redness or swelling at spot shot was given
- Headache
- Fever
- Fatigue
- Irritability, diarrhea or loss of appetite in healthy infants and children (Recombivax, Vaxelis, Pediarix)
- Vomiting, crying or drowsiness in children (Vaxelis, Pediarix)
Severe allergic reactions to hep B vaccine are rare but can be life threatening. In the event of a severe allergic reaction, call 9-1-1 and get the person to the closest hospital.
Who Should Not Get Hep B Vaccine
People should not get hep B vaccine if they:
- Have had a severe allergic reaction to a past dose of hep B vaccine or to any vaccine component
- Have a yeast allergy (PREHEVBRIO is the the only hep vaccine that does not contain yeast and is safe for people with a yeast allergy)
- Have had an allergic reaction to neomycin (if they are getting Twinrix)
- Are pregnant (if they are getting Heplisav-B)
People with a minor illness such as a cold can get hep B vaccine but people with a moderate or severe illness should usually wait to get hep B vaccine until after they recover.
Talk to your health care provider about whether or not hep B vaccine is right for you.
Available Vaccines
One vaccine that protects against human papillomavirus is approved in the U.S. For HPV vaccine to work best, vaccine series should start before being exposed to HPV.
Who Should Get HPV Vaccine
Two or three doses of hep B are recommended by CDC. Children who start HPV vaccine series, ideally at age 11-12 years, should get two doses at least six months apart. Adolescents who get two doses less than five months apart and those age 15-26 years should get three doses. People age 27-45 years may get HPV vaccine after speaking with their health care provider.
Common Side Effects
HPV vaccine is safe and the best way to prevent HPV-related cancers and infections. Like any medicine, vaccines can have side effects. The side effects are usually mild and go away on their own.
Common side effects of HPV vaccine include:
- Pain, soreness, redness or swelling at spot shot was given
- Fever
- Headache or feeling tired
- Nausea
- Muscle or joint pain
Severe allergic reactions to HPV vaccine are rare but can be life threatening. In the event of a severe allergic reaction, call 9-1-1 and get the person to the closest hospital.
Who Should Not Get HPV Vaccine
People should not get HPV vaccine if they:
- Have had a severe allergic reaction to a past dose of HPV vaccine or to any vaccine component
- Are pregnant
HPV vaccine is not recommended for people who are pregnant but a person should not be alarmed if they get HPV vaccine while pregnant. If a person finds they are pregnant after starting HPV vaccine series they should wait to get second and/or third doses until they are no longer pregnant. People who are breastfeeding can get HPV vaccine.
Talk to your health care provider about whether or not HPV vaccine is right for you.
Available Vaccines
There are two ways to get flu vaccine approved in the U.S. - by injection (a shot) or nasal spray. One flu vaccine is not recommended over another. In any flu season, multiple manufacturers have FDA-approved flu vaccines.
Who Should Get Flu Vaccine
Annual flu vaccine is recommended by CDC for all people age 6 months and older. People at high risk for complications from influenza include:
Common Side Effects
Flu vaccine is safe and the best way to prevent influenza. Like any medicine, vaccines can have side effects. The side effects are usually mild and go away on their own.
Common side effects of injection flu vaccine include:
- Soreness, redness or swelling at spot shot was given
- Fever
- Muscle aches
- Headache
- Fatigue
Common side effects of nasal spray flue vaccine in children include:
- Runny nose
- Wheezing
- Headache
- Vomiting
- Muscle aches
- Low grade fever
Common side effects of nasal spray flue vaccine in adults include:
- Runny nose
- Headache
- Sore throat
- Cough
Severe allergic reactions to flu vaccine are rare but can be life threatening. In the event of a severe allergic reaction, call 9-1-1 and get the person to the closest hospital.
Who Should Not Get Flu Vaccine
People should not get injection flu vaccine if they:
- Have had a severe allergic reaction to a past dose of injection flu vaccine or to any vaccine component
- Are younger than age 6 months
People should talk to their health care provider before getting injection flu vaccine if they:
- Have an allergy to eggs or any vaccine ingredients
- Have ever had GBS (Guillain-Barré Syndrome)
People should not get nasal spray flu vaccine if they:
- Have had a severe allergic reaction to a past dose of nasal spray flu vaccine or any vaccine component
- Are younger than age 2 years
- Are age 50 years or older
- Are pregnant
- Are between ages 2-17 years and are receiving aspirin, aspirin-containing or salicylate-containing products
- Have weakened immune systems
- Are close contacts of or care for people who are severely immunocompromised and require a protected environment (or avoid contact with those person for seven days after getting nasal spray flu vaccine)
- Are age 2-4 years and have asthma or a history of wheezing in the past 12 months
- Have certain chronic health conditions
People should talk to their health care provider before getting nasal spray flu vaccine if they:
- Have recently taken influenza antiviral drugs
- Are age 5 years and older and have asthma
- Have underlying medical conditions that can increase their risk of serious flu complications
- Have moderate or severe illness with or without fever
- Have had GBS (Guillain-Barré Syndrome) after a past dose of flu vaccine
People who are moderately or severely sick should usually wait to get flu vaccine until they recover.
Talk to your health care provider about whether or not flu vaccine is right for you.
Available Vaccines
There are two MMR vaccines approved in the U.S. Both are approved for people age 12 months and older. MMRV vaccine also protects against measles, mumps and rubella.
Who Should Get MMR Vaccine
Two doses of MMR vaccine is recommended by CDC. Children should get their first dose of MMR vaccine at age 12 months and their second at age 4-6 years.
Common Side Effects
MMR vaccine is safe and the best way (along with MMRV vaccine) to prevent measles, mumps and rubella. Like any medicine, vaccines can have side effects. The side effects are usually mild and go away on their own.
Common side effects of MMR vaccine include:
- Sore arm from shot
- Fever
- Mild rash
- Temporary stiffness and pain in joints (mostly in teenage and adult women without prior immunity to the vaccine's rubella component)
MMR vaccine has a very small risk of febrile seizures for infants. This risk increases as infants get older, so it is recommended for infants to get MMR vaccine as soon as they are old enough.
MMR vaccine can cause some people's cheeks or necks to swell. MMR vaccine can also cause a temporary low platelet count that is not life threatening and usually goes away without treatment.
Severe allergic reactions to MMR vaccine are rare but can be life threatening. In the event of a severe allergic reaction, call 9-1-1 and get the person to the closest hospital.
Who Should Not Get MMR Vaccine
People should not get MMR vaccine if they:
- Have had a severe allergic reaction to a past dose of MMR vaccine or to any vaccine component
- Have ever had a life-threatening allergic reaction to the antibiotic neomycin
People with a minor illness such as a cold can get MMR vaccine but people with a moderate or severe illness should usually wait to get MMR vaccine until after they recover.
Talk to your health care provider about whether or not MMR vaccine is right for you.
Available Vaccines
There is one MMRV vaccine approved in the U.S. It is approved for people age 1-12 years. MMR vaccine also protects against measles, mumps and rubella.
Who Should Get MMRV Vaccine
MMRV vaccine is recommended by CDC for all children age 1-12 years.
Common Side Effects
MMRV vaccine is safe and the best way (along with MMRV vaccine) to prevent measles, mumps, rubella and varicella (chickenpox). Like any medicine, vaccines can have side effects. The side effects are usually mild and go away on their own.
Common side effects of MMRV vaccine include:
- Sore arm from shot
- Fever
- Mild rash
MMRV vaccine has a very small risk of febrile seizures for infants. This risk increases as infants get older, so it is recommended for infants to get MMRV vaccine as soon as they are old enough.
MMRV vaccine can cause some people's cheeks or necks to swell. MMR vaccine can also cause a temporary low platelet count that is not life threatening and usually goes away without treatment.
Severe allergic reactions to MMRV vaccine are rare but can be life threatening. In the event of a severe allergic reaction, call 9-1-1 and get the person to the closest hospital.
Who Should Not Get MMRV Vaccine
People should not get MMRV vaccine if they:
- Have had a severe allergic reaction to a past dose of MMRV vaccine or to any vaccine component
- Have ever had a life-threatening allergic reaction to the antibiotic neomycin
People with a minor illness such as a cold can get MMRV vaccine but people with a moderate or severe illness should usually wait to get MMRV vaccine until after they recover.
Talk to your health care provider about whether or not MMRV vaccine is right for you.
Available Vaccines
There are two types of meningococcal vaccine approved in the U.S. There is one conjugate vaccine that protects against meningococcal caused by serogroups A, C, W and Y (MenACWY vaccine) and two that protect against meningococcal caused by serogroup B (MenB vaccine).
Who Should Get MenACWY/MenB Vaccine
Two doses of MenACWY vaccine are recommended by CDC. Adolescents should get their first dose of MenACWY vaccine at age 11-12 years and their second at age 16 years. MenACWY vaccine is also recommended for some people age 2 months and older.
Multiple doses of MenB vaccine are recommended by CDC for people age 16-18 years and some people age 10 years and older.
People with a minor illness such as a cold can get MenACWY and MenB vaccines but people with a moderate or severe illness should usually wait to get MenACWY and MenB vaccines until after they recover.
Common Side Effects
MenACWY vaccine is safe and the best way to protect against meningicoccal caused by serogroups A, C, W and Y. Like any medicine, vaccines can have side effects. The side effects are usually mild and go away on their own.
Common side effects of MenACWY vaccine are mild to moderate, last 1-3 days and include:
- Soreness, redness or swelling at spot shot was given
- Muscle pain
- Headache
- Fatigue
MenB vaccine is safe and the best way to protect against meningococal caused by serogroup B. Like any medicine, vaccines can have side effects. The side effects are usually mild and go away on their own.
Common side effects of MenB Vaccine mild to moderate, last 1-3 days and include:
- Soreness, redness or swelling at spot shot was given
- Headache
- Fatigue
- Joint or muscle pain
- Fever
Severe allergic reactions to MenACWY and MenB vaccines are rare but can be life threatening. In the event of a severe allergic reaction, call 9-1-1 and get the person to the closest hospital.
Who Should Not Get MenACWY/MenB Vaccine
People should not get MenACWY vaccine if they:
- Have had a severe allergic reaction to a past dose of MenACWY vaccine or to any vaccine component
- Have had a severe allergic reaction after a past dose of diphtheria toxoid-, CRM or tetanus toxoid-containing vaccine
- Have any severe, life-threatening allergies
People should not get MenB vaccine if they:
- Have had a severe allergic reaction to a past dose of MenACWY vaccine or to any vaccine component
- Have any severe, life-threatening allergies
- Are pregnant or breastfeeding
- Have a latex allergy (and are getting Bexsero)
Talk to your health care provider about whether or not MenACWY/MenB vaccine is right for you.
Available Vaccines
There are two types of mpox vaccine approved in the U.S. There is one replication-deficient modified vaccinia Ankara (MVA) vaccine and one replication-competent vaccinia virus vaccine.
Who Should Get Mpox Vaccine
Factors related to who is recommended by CDC to get Mpox vaccine are changing due to the 2022 mpox outbreak.
Common Side Effects
Mpox vaccine is safe and the best way to protect against mpox. Like any medicine, vaccines can have side effects. The side effects are usually mild and go away on their own.
Common side effects of mpox vaccine include:
- Itching
- Swollen lymph nodes
- Sore arm
- Fever
- Headache
- Body ache
- Mild rash
- Fatigue
Severe allergic reactions to mpox vaccine are rare but can be life threatening. In the event of a severe allergic reaction, call 9-1-1 and get the person to the closest hospital.
Who Should Not Get Mpox Vaccine
People should not get mpox vaccine if they:
- Have had a severe allergic reaction to a past dose of mpox vaccine or to any vaccine component
- Have had a severe allergic reaction after getting gentamicin or ciprofloxacin
- Have had a severe allergic reaction to egg or chicken protein AND are currently avoiding exposure to egg or chicken proteins
Talk to your health care provider about whether or not mpox vaccine is right for you.
Available Vaccines
There are four pneumococcal vaccines approved in the U.S. Different pneumococcal vaccines are recommended for different people.
Who Should Get Pneumococcal Vaccine
Four or more doses of pneumococcal vaccine is recommended for all children under age 5 years, all people ages 5-64 years at a higher risk for pneumococcal disease and all adults age 65 years and older.
Common Side Effects
Pneumococcal vaccine is safe and the best way to protect against pneumococcal disease. Like any medicine, vaccines can have side effects. The side effects are usually mild and go away on their own.
Common side effects of pneumococcal vaccines include:
- Feeling drowsy
- Loss of appetite
- Swollen or sore arm from shot
- Fever
- Headache
Severe allergic reactions to pneumococcal vaccine are rare but can be life threatening. In the event of a severe allergic reaction, call 9-1-1 and get the person to the closest hospital.
Who Should Not Get Pneumococcal Vaccine
People should not get pneumococcal vaccine if they:
- Have had a severe allergic reaction to a past dose of pneumococcal vaccine or to any vaccine component
- Have had any severe, life-threatening allergies
- Are under age 2 years
People with a minor illness such as a cold can probably get pneumococcal vaccine but people with a moderate or severe illness should probably wait to get pneumococcal vaccine until after they recover.
Talk to your health care provider about whether or not pneumococcal vaccine is right for you.
Available Vaccines
There are two rotavirus vaccines approved in the U.S. Rotarix is approved for infants ages 6-24 weeks and RotaTeq is approved for infants ages 6-32 weeks.
Who Should Get Rotavirus Vaccine
Two or three doses of rotavirus vaccine are recommended by CDC depending on brand. Infants should get their first dose at age 2 months, second at age 4 months and third (depending on brand) at age 6 months.
Infants with a minor illness such as a cold can get rotavirus vaccine but people with a moderate or severe illness should usually wait to get rotavirus vaccine until after they recover.
Common Side Effects
Rotavirus vaccine is safe and the best way to protect against rotavirus. Like any medicine, vaccines can have side effects. The side effects are usually mild and go away on their own.
Common side effects rotavirus vaccine include:
- Irritability
- Mild, temporary diarrhea or vomiting
Some studies suggest a link between rotavirus vaccine and a very rare, small increase in risk of intussusception, a type of bowel blockage.
Severe allergic reactions to rotavirus vaccine are rare but can be life threatening. In the event of a severe allergic reaction, call 9-1-1 and get the person to the closest hospital.
Who Should Not Get Rotavirus Vaccine
Infants should not get rotavirus vaccine if they:
- Have had a severe allergic reaction to a past dose of rotavirus vaccine or to any vaccine component
- Have a weakened immune system
- Have SCID (severe combined immunodeficiency)
- Have had intussusception (a type of bowel blockage)
Talk to your health care provider about whether or not rotavirus vaccine is right for you.
Available Vaccines
There is one shingles vaccines approved in the U.S. In the past Zostavax was also available, but Shingrix was recommended over Zostavax by CDC for stronger protection against shingles. Shingrix is still available in the U.S.
Who Should Get Shingles Vaccine
Two or three doses of rotavirus vaccine are recommended by CDC depending on brand. Infants should get their first dose at age 2 months, second at age 4 months and third (depending on brand) at age 6 months.
People with a minor illness such as a cold can get rotavirus vaccine but people with a moderate or severe illness should usually wait to get rotavirus vaccine until after they recover.
Common Side Effects
Shingles vaccine is safe and the best way to protect against shingles (herpes zoster). Like any medicine, vaccines can have side effects. The side effects are usually mild and go away on their own.
Common side effects of shingles vaccine are mild to moderate, last 2-3 days, are more common in younger people and include:
- Pain, redness and swelling at spot shot was given
- Muscle pain
- Tiredness
- Headache
- Shivering
- Fever
- Upset stomach
Severe allergic reactions to shingles vaccine are rare but can be life threatening. In the event of a severe allergic reaction, call 9-1-1 and get the person to the closest hospital.
Who Should Not Get Shingles Vaccine
People should not get shingles vaccine if they:
- Have had a severe allergic reaction to a past dose of shingles vaccine or to any vaccine component
- Have shingles
- Are pregnant or breastfeeding
Talk to your health care provider about whether or not shingles vaccine is right for you.
Smallpox Vaccine
Because smallpox has been eradicated, routine smallpox vaccine for the general public has stopped. CDC recommends smallpox vaccine only for people at risk for occupational exposure to orthopoxviruses. The U.S. government has stockpiled enough smallpox vaccine for all people in the U.S. to get smallpox vaccine as a safety measure for if a smallpox outbreak occurs due to bioterrorism.
Additional Resources
- Common Vaccine Safety Questions and Concerns
- Vaccines During Pregnancy FAQs
- Vaccine Safety Datalink (VSD)
- Vaccine Safety Information for Parents and Caregivers
- Vaccine Safety Information for Health Care Providers
- Emergency Preparedness and Vaccine Safety
- Common Questions About Vaccines
- Vaccine Education Center at Children's Hospital of Philadelphia
Questions? Contact Us!
NDHHS Disease Control — Immunization Unit
600 E. Boulevard Avenue, Dept. 325
Bismarck, ND 58505-0200
Phone: (701) 328-3386
Toll-Free: (800) 472-2180
TTY: 711
Email: vaccine@nd.gov